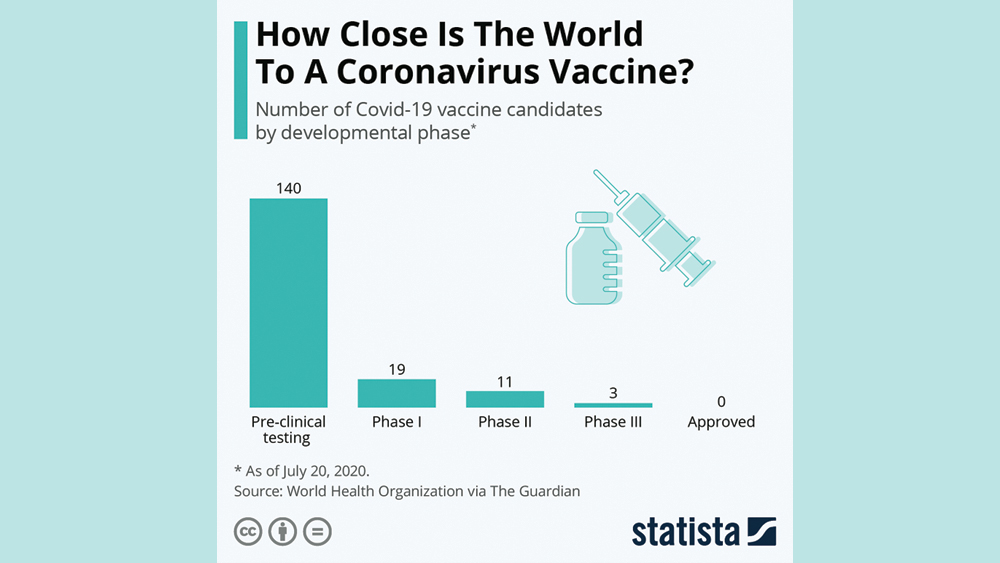
The Lancet, a scientific journal, published the hugely promising results of Phase I/II trials and they’ve raised hopes that a safe, effective and accessible vaccine is attainable to help end the pandemic. The trial provoked a T cell response within 14 days of vaccination and an antibody response within 28 days. Before testing began, there were commitments to supply more than 2 billion doses of the vaccine to the U.S., the United Kingdom, Europe’s Inclusive Vaccines Alliance, the Coalition of Epidemic Preparedness, Gavi the Vaccine Alliance and the Serum Institute of India. Vaccines generally take years to develop, but the University of Oxford has been working on its current project at an unprecedented pace. It’s one of more than 140 COVID-19 vaccine candidates listed by the World Health Organization, according to The Guardian (a British newspaper) and several are already in advanced testing.